 Sister-chromatid cohesion mediated by the alternative RF-CCtf18/Dcc1/Ctf8, the helicase Chl1 and the polymerase-α-associated protein Ctf4 is essential for chromatid disjunction during meiosis II. Replisome progression complex links DNA replication to sister chromatid cohesion in Xenopus egg extracts. mcl1+, the Schizosaccharomyces pombe homologue of CTF4, is important for chromosome replication, cohesion, and segregation. Influence of the human cohesion establishment factor Ctf4/AND-1 on DNA replication. Drosophila Ctf4 is essential for efficient DNA replication and normal cell cycle progression. Evidence that POB1, a Saccharomyces cerevisiae protein that binds to DNA polymerase α, acts in DNA metabolism in vivo. 
CTF4 (CHL15) mutants exhibit defective DNA metabolism in the yeast Saccharomyces cerevisiae. Mcm10 and And-1/CTF4 recruit DNA polymerase alpha to chromatin for initiation of DNA replication. Mrc1 and DNA polymerase ε function together in linking DNA replication and the S phase checkpoint. 
Dpb2 integrates the leading-strand DNA polymerase into the eukaryotic replisome. A key role for Ctf4 in coupling the MCM2–7 helicase to DNA polymerase α within the eukaryotic replisome. Ctf4 coordinates the progression of helicase and DNA polymerase α. Stoichiometry and architecture of active DNA replication machinery in Escherichia coli. Characterization of a triple DNA polymerase replisome. Single-molecule studies reveal the function of a third polymerase in the replisome. Polymerase exchange during Okazaki fragment synthesis observed in living cells. Mechanisms of replication fork protection: a safeguard for genome stability. Unwind and slow down: checkpoint activation by helicase and polymerase uncoupling. DNA replication as a target of the DNA damage checkpoint. Genome Duplication (Garland Science, 2011) The ability of Ctf4 to act as a platform for multivalent interactions illustrates a mechanism for the concurrent recruitment of factors that act together at the fork.ĭePamphilis, M. Our findings indicate that Ctf4 can couple two molecules of Pol α to one CMG helicase within the replisome, providing a new model for lagging-strand synthesis in eukaryotes that resembles the emerging model for the simpler replisome of Escherichia coli 5, 6, 7, 8. Accordingly, we demonstrate that one Ctf4 trimer can support binding of up to three partner proteins, including the simultaneous association with both Pol α and GINS. We show that the amino-terminal tails of the catalytic subunit of Pol α and the Sld5 subunit of GINS contain a conserved Ctf4-binding motif that docks onto the exposed helical extension of a Ctf4 protomer within the trimer. 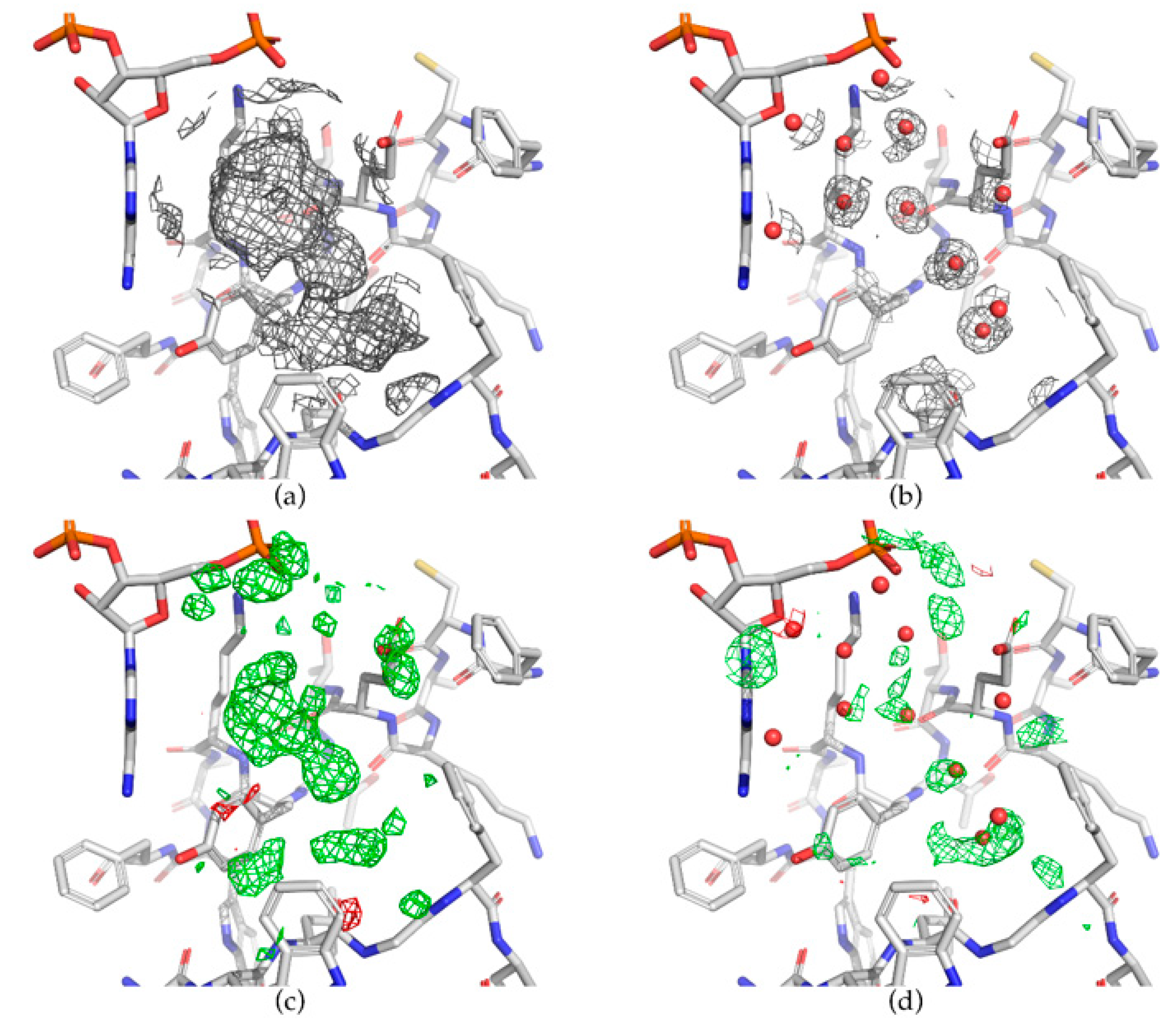
Critically, Pol α and the CMG helicase share a common mechanism of interaction with Ctf4. Trimerization depends on a β-propeller domain in the carboxy-terminal half of the protein, which is fused to a helical extension that protrudes from one face of the trimeric disk. We use X-ray crystallography and electron microscopy to show that Ctf4 self-associates in a constitutive disk-shaped trimer. Here we define the molecular mechanism by which the yeast Ctf4 protein links the Cdc45–MCM–GINS (CMG) DNA helicase to DNA polymerase α (Pol α) within the replisome. In eukaryotes, the physical coupling between helicase and DNA polymerases remains poorly understood. Efficient duplication of the genome requires the concerted action of helicase and DNA polymerases at replication forks 1 to avoid stalling of the replication machinery and consequent genomic instability 2, 3, 4. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
